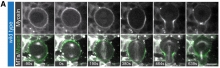
The Cabernard lab and collaborators recently published in Developmental Cell and found that cell and tissue morphogenesis depends on the correct regulation of non-muscle Myosin II, but how this motor protein is spatiotemporally controlled is incompletely understood. They show that in asymmetrically dividing Drosophila neural stem cells, cell intrinsic polarity cues provide spatial and temporal information to regulate biased Myosin activity. Using live cell imaging and a genetically encoded Myosin activity sensor, they found that Drosophila Rho kinase (Rok) enriches for activated Myosin on the neuroblast cortex prior to nuclear envelope breakdown (NEB). After NEB, the conserved polarity protein Partner of Inscuteable (Pins) sequentially enriches Rok and Protein Kinase N (Pkn) on the apical neuroblast cortex. Their data suggest that apical Rok first increases phospho-Myosin, followed by Pkn-mediated Myosin downregulation, possibly through Rok inhibition. They propose that polarity-induced spatiotemporal control of Rok and Pkn is important for unequal cortical expansion, ensuring correct cleavage furrow positioning and the establishment of physical asymmetry.
Read the full article in Developmental Cell